Toronto, Ontario--(Newsfile Corp. - March 17, 2026) - Theralase® Technologies Inc. (TSXV: TLT) (OTCQB: TLTFF) ("Theralase®" or the "Company"), a clinical stage pharmaceutical company, dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses, is pleased to announce the discovery of an additional Mechanism Of Action ("MOA") as to how Ruvidar® is so effective in the inactivation of the Herpes Simplex Virus ("HSV-1").
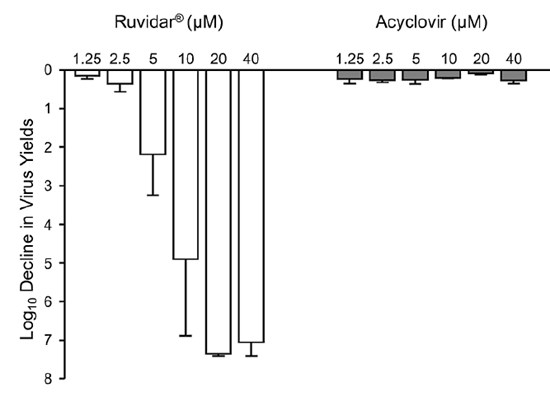
Theralase® previously reported that Ruvidar® (TLD-1433) was effective in the inactivation of HSV-1 and was superior to the standard of care treatment Acyclovir. See Figure 1.1,2,3 (April 10, 2025 - "Ruvidar More Effective in the Treatment of Herpes than FDA-Approved Treatments" and September 24, 2025 - "Independent Research Demonstrates Ruvidar® Effective in the Destruction of Herpes Simplex Virus")
Figure 1: Anti-viral effect of Ruvidar® versus Acyclovir in HSV-1 (24 hours post infection)
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/288729_775fcea0a401881b_001full.jpg
To more fully understand the MOA of how Ruvidar® is so effective in the inactivation of HSV-1, Theralase®'s research team, further investigated the ability of Ruvidar® to inactivate the virus and protect host cells.
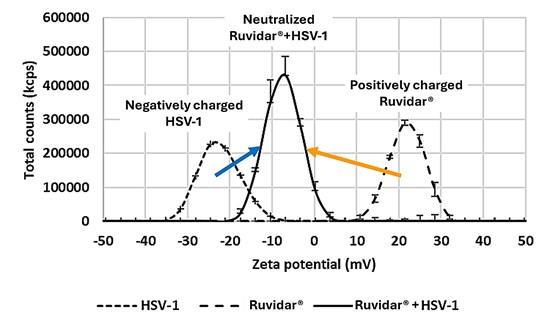
An additional MOA to be investigated was the electrostatic attraction between positively charged Ruvidar® and negatively charged HSV-1. This interaction was easily validated by measuring the zeta potential (reflects surface charge) in aqueous solutions of Ruvidar®, HSV-1 and in a combination of both. A reversal or significant change in the zeta potential of the combination would confirm the electrostatic binding of Ruvidar® to HSV-1.
The results of this experiment are shown in Figure 2.
Figure 2: Binding of Ruvidar® to HSV-1
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/288729_775fcea0a401881b_002full.jpg
As shown in Figure 2, Ruvidar® effectively binds to HSV-1, neutralizing the surface charge of the virus, rendering it unable to infiltrate cells and halting the proliferation of the virus.
By significantly reducing the negative charge of HSV-1, the virus is rendered non-infectious preventing its ability to bind and infect human cells.
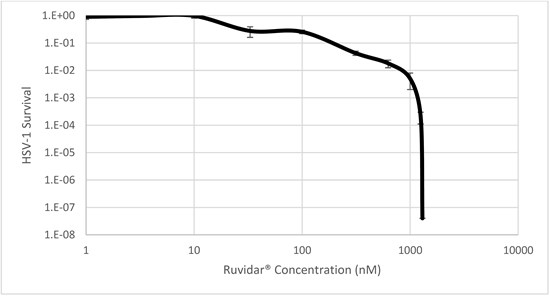
Figure 3: Inactivation of HSV-1 by Ruvidar® in vitro
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/288729_775fcea0a401881b_0003full.jpg
To support this discovery, Figure 3 demonstrates that Ruvidar® in the micromolar range (1,300 nM = 1.3 µM) is able to completely destroy HSV-1, providing up to an 8 log reduction in magnitude.
To fully understand, an 8 log reduction in magnitude represents a 99.999999% reduction in the virion (complete infective virus) population. This means that for a population of 100,000,000 virions, only 1 virion or less is expected to survive.
Pavel Kaspler, PhD, Research Scientist, Theralase®, who conducted the study, stated: "This latest preclinical study has demonstrated that Ruvidar® is very effective in inactivating HSV-1 by binding to the virus and neutralizing its negative charge. This, combined with its ability to stop the spread of infection in cells; thus, preventing virus transmission, points to a highly promising, clinically beneficial treatment for the herpes simplex virus".
Arkady Mandel, MD, PhD, DSc, Chief Scientific Officer at Theralase®, stated that "Theralase®'s latest research offers valuable new insight into how Ruvidar® can help fight viral infections, which would be of preeminent significance to the billions of people infected by viruses every year. The experiments conducted by Dr. Coombs and our research team support the ideology that Ruvidar® is able to attack viruses on multiple fronts: interrupting the 'virus life cycle'; thus, preventing the virus from instructing the host cell to replicate its genetic material and produce new progeny virions, destroying intracellular viruses through the production of singlet oxygen, inhibiting critical intracellular processes required for virus survival and affecting enzymes linked to pathogenicity of the virus".
Dr. Mandel noted that "We can now add to this list that Ruvidar® is able to bind to negatively charged components in the virus; including: envelopes and capsids, thus preventing the virus from entering cells and interfering with the stages of uncoating and genome release. Furthermore, Ruvidar® is believed to block viruses from entering the cell's nucleus, thus stopping them from replicating. Ruvidar® may also bind directly to viral genomes, disrupting replication. This research strongly supports the clinical development of Ruvidar® as an effective antiviral therapy".
Roger DuMoulin-White, B.Sc., P.Eng., Pro.Dir., President and Chief Executive Officer, Theralase® stated, "This latest research provides even further evidence of the MOA of Ruvidar® in the inactivation of HSV-1. Preclinical and clinical development for a topical HSV-1 treatment is slated to commence in 2026."
About Herpes Simplex:
Herpes Simplex Virus ("HSV"), known as herpes, is a very common infection that can cause painful blisters or ulcers on the skin of an individual. It primarily spreads by skin-to-skin contact, while it is treatable, it is not curable.4
There are two main types of HSV:4
Type 1 ("HSV-1") generally spreads by oral contact and causes infections in or around the mouth, vermilion, upper or lower lip region (oral herpes or cold sores). It can also cause genital herpes. A majority of adults are infected with HSV-1.
Type 2 ("HSV-2") spreads by sexual contact and causes herpes in the genital region of an individual.
An estimated 3.8 billion people under the age of 50 (64%) globally have HSV-1, the main cause of oral herpes. An estimated 520 million people aged 15 to 49 (13%) globally have HSV-2, the main cause of genital herpes.4
The global HSV treatment market size was estimated at $USD 2.8 billion in 2024 and is expected to balloon to $USD 4.7 billion by 2033.5
References:
1 Coombs K.M., Glover K.K.M., Russell R., Kaspler P., Roufaiel M., Graves D., Pelka P., Kobasa D., DuMoulin-White R., Mandel A. Nanomolar concentrations of the photodynamic compound TLD-1433 effectively inactivate numerous human pathogenic viruses. Heliyon. 2024 May 29;10(11):e32140.
2 Coombs K.M., DuMoulin-White R., Mandel A. Ruvidar®-An Effective Anti-Herpes Simplex Virus Agent. Viruses. 2025 Sep 20;17(9):1280.
3 Ruvidar More Effective in the Treatment of Herpes than FDA-Approved Treatments - Theralase Technologies
4 Herpes simplex virus
5 Herpes Simplex Virus Treatment Market Size, Top Share, Key Developments | By 2033
About Theralase® Technologies Inc.:
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses.
Additional information is available at www.theralase.com and www.sedarplus.ca.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains Forward-Looking Statements ("FLS") within the meaning of applicable Canadian securities laws. Such statements include; but, are not limited to statements regarding the Company's proposed development plans with respect to small molecules and their drug formulations. FLS may be identified by the use of the words "may, "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of the Company's management regarding future research, development and commercialization of the Company's small molecules; their drug formulations; preclinical research; clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to fund and secure regulatory approvals to successfully complete various clinical studies in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its small molecule and drug formulations; access to sufficient capital to fund the Company's operations is available on terms that are commercially favorable to the Company or at all; the Company's small molecule and formulations may not be effective against the diseases tested in its clinical studies; the Company fails to comply with the terms of license agreements with third parties and as a result loses the right to use key intellectual property in its business; the Company's ability to protect its intellectual property; the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these FLS, which are not a guarantee of future performance. There can be no assurance that FLS will prove to be accurate as such FLS involve known and unknown risks, uncertainties and other factors, which may cause actual results or future events to differ materially from the FLS.
Although the FLS contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these FLS.
All FLS are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such FLS.
For investor information on the Company, please feel to reach out Investor Inquiries - Theralase Technologies.
For More Information:
1.866.THE.LASE (843.5273)
416.699.LASE (5273)
www.theralase.com
Kristina Hachey, CPA
Chief Financial Officer X 224
khachey@theralase.com
To view the source version of this press release, please visit https://www.newsfilecorp.com/release/288729