FAYETTEVILLE, Ark. - Dec. 21, 2021 - PRLog -- After receiving FDA clearance in May 2021 for its flagship product – SafeBreak Vascular, Lineus Medical continues to expand its distribution network with the addition of Alamo Scientific, a specialty medical distributor based out of San Antonio, Texas. Alamo Scientific services hospitals in Texas, Oklahoma, Southern Nevada, and New Mexico on behalf of Lineus Medical. They employ 4 salespeople covering this geographic territory and have a total of 10 employees.
"We have partnered with another strong and respected vascular access distribution and sales company to cover an important territory in the United States," Vance Clement, CEO of Lineus Medical stated. "Alamo Scientific is well represented in Texas where almost 9% of the US population lives. Their product portfolio compliments SafeBreak Vascular perfectly, and with their large customer base and strong personal relationships within this area, we expect great things from this partnership."
"Alamo Scientific is excited to be a partner with Lineus Medical in distributing SafeBreak Vascular in Texas and the surrounding states," said the president of Alamo Scientific, Todd Endersby. "SafeBreak Vascular fits nicely into our innovative product portfolio that focuses on improving healthcare on the frontlines."
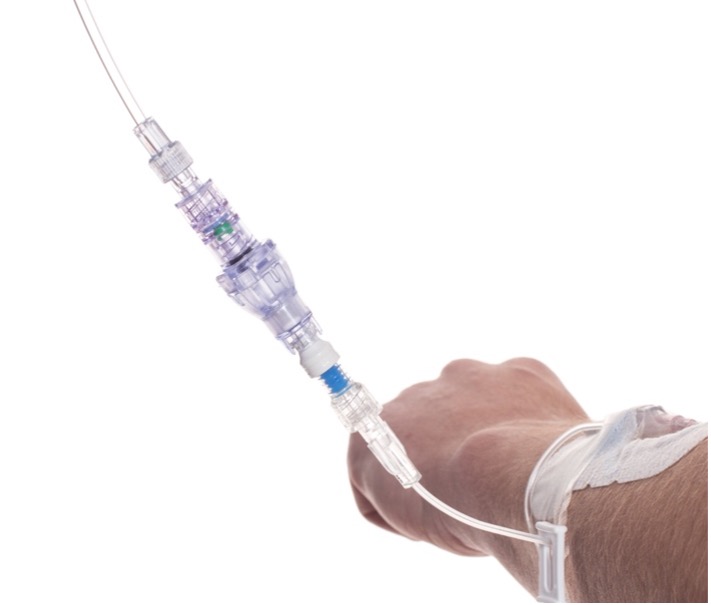
SafeBreak Vascular is a break-away connector that fits into any peripheral IV line on any standard luer lock in the world. The device is designed to separate when damaging force is placed on an IV line, reducing irritation of the vein and the chances of the IV dislodging. When the device separates, valves on each side of the device close to stop the flow of medication from the pump and blood flow from the patient's IV catheter. The separated SafeBreak can be replaced with a new, sterile SafeBreak and the IV restarted without the need for another needlestick for the patient. In a 2021 randomized controlled trial, SafeBreak Vascular was shown to reduce IV complications requiring an IV restart by 44%.1
SafeBreak Vascular reduces the failure of peripheral IV lines and the negative cascade of events that occurs when an IV restart is required. Peripheral IVs fail 46% of the time before their intended use is complete according to the clinical literature and cost the US healthcare system over $5B annually2.
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and other types of medical tubing may be reduced when a force-activated separation device separates to protect the patient. Learn more at www.lineusmed.com and follow us on LinkedIn or Twitter.
1Data on file.
2Helm, R.E., et al., Accepted but Unacceptable: Peripheral IV Catheter Failure. Journal of Infusion Nursing. 2015;38(3): 189-203
MKG-0121 Rev 00
Contact
Vance Clement
***@lineusmed.com
Photos: (Click photo to enlarge)![]()

![]()
Read Full Story - Lineus Medical Goes Big in Texas with Alamo Scientific, Expands Distribution Network in the Southwestern United States | More news from this source
Press release distribution by PRLog
Lineus Medical Goes Big in Texas with Alamo Scientific, Expands Distribution Network in the Southwestern United States
December 21, 2021 at 08:30 AM EST


